Institute for Protein Physics
Topics (continued)
 |
|
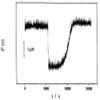
(T.7) 25th Anniversary of the Publication of the First Paper on Enzyme Activity Assessment Using Isothermal Titration Calorimetry (ITC) |
|
This paper, published 25 years ago on February 2, 2001, reported that the catalytic hydrolysis and condensation reactions of the industrial enzyme thermolysin could be quantitatively observed by utilizing the feature of calorimetry, which allows direct measurement of reaction rates. Furthermore, it was demonstrated that the steady-state observables for the entire reaction process, particularly in hydrolysis reactions, can be well explained by a steady-state model of the Michaelis-Menten mechanism. This made it possible to determine the apparent enzyme reaction parameters (turnover number and Michaelis constant) from a single measurement of a single substrate concentration. This method has since been applied to various enzyme reactions. In particular, it was shown that for many enzymes, the apparent Michaelis constant changes linearly with the initial substrate concentration when the initial substrate concentration is changed, indicating competitive product inhibition. An overview of this method is also presented in the tutorial series "Calorimetry of Protein Molecules(4)" (Netsu Sokutei, Vol. 53, No. 1, pp. 29-30, 2026) in the journal of Japan Society of Calorimetry and Thermal Analysis.
|
